Treatment options
In this section you will find information about brain tumour treatment options. Decisions will not be easy, and you should be fully involved in them. The more you know the better – this section should answer your questions, and raise more for you to discuss with your clinical team. Do your research so you can be empowered to take control of your treatment and decide what the right path is for you. Ask, ask and ask again. And you can ask us.
Therapies
Having a brain tumour is complex. Treating a brain tumour is complex. Increasingly, brain tumour treatment options are being ‘personalised’ to meet the needs of the patient.
Today, clinicians no longer treat just plain cancer; they use the knowledge of the biology of cancer to plan treatments more effectively, as they know so much more about it. The current options include surgery, radiation therapy, and chemotherapy (chemo). Having ‘chemo’ will probably conjure up an image of a terribly sick, bald person. There have been major improvements in chemotherapy so that most regimens are well tolerated.
Many people have a combination of treatments and the choice of treatment depends mainly on the following
- The type and grade of brain tumour
- Its location in the brain
- Its size
- Age
- General health
Cancer therapy often damages healthy cells and tissues and therefore side effects are common. Before treatment starts, ask your oncologist about possible side effects and how treatment may change your normal activities.
These are things you may want to ask before you begin treatment:
- What are my treatment choices? Are there any choices available elsewhere that aren’t available here?
- Which do you recommend for me? Why? How does that treatment work?
- What are the expected benefits of each kind of treatment?
- What can I do to prepare for treatment?
- Will I need to stay in the hospital? If so, for how long?
- What are the risks and possible side effects of each treatment? How can side effects be managed?
- How will treatment affect my normal activities? What is the chance that I will have to learn how to walk, speak, read, or write after treatment?
- Would a research study (clinical trial) be appropriate for me? If it isn’t, why not?
- I might decide to seek a second opinion. What would the questions be that you would ask?
Explore therapies
In this section, you will can more information about the different types of therapies that are used to treat brain tumours.
Brain surgery
When you are diagnosed with a brain tumour, one of the treatment options may be neurosurgery. On this page you will find the information you need to help you feel as prepared and in control as possible.
Neurosurgery
In this section you will find out more information about:
Molecular analysis
Will chemotherapy work on my brain tumour? This is a question we hear a lot, and is in important one in understanding what brain tumour treatment might work best for you. Molecular analysis can help answer this question.
Such is the progress in personalised treatments that neuropathologists can now identify which tumours are likely to respond to chemotherapy. Specific tests might provide information that can be used to influence your treatment and diagnosis. Not all hospitals run such tests but you can ask for them. These tests (also called molecular markers) can:
- Aid the diagnosis of brain tumours which are sometimes hard to diagnose
- Allow clinicians to work out a prognosis
- Indicate whether a tumour will respond to a specific type of treatment
The downside of this is that molecular analysis may lead you to discover that your tumour type might not respond to treatment, so if you ask the question, you need to be prepared that you might receive information you’d rather not know.
MGMT methylation testing and brain tumours
The MGMT methylation test detects a chemical change in the DNA that shows how the cells are able to fight certain chemotherapy drugs (i.e. repair the damage caused by the drug so that the cancer cells survive and continue to grow). Tumour samples can also be tested for a ‘1p/19q’ genetic change in the chromosomes that may provide information about the likely sensitivity to DNA acting drugs.
The MGMT and 1p/19q tests will only be relevant to some brain tumours. They can also only be done where a biopsy has been performed, and biopsy material can be obtained and analysed (it does not necessarily matter how long ago the biopsy was performed).
The MGMT methylation test is relevant to all anaplastic gliomas (WHO Grades III and IV).
1p /19q test is relevant to certain tumour types.
The tumour types for which these tests can be done are shown by a tick below.
| Brain tumour diagnosis | Grade | MGMT | 1p/19q |
| Anaplastic astrocytoma | WHO grade III | ✓ | ✗ |
| Oligodendroglioma | WHO grade II | ✗ | ✓ |
| Anaplastic Oligodendroglioma | WHO grade III | ✓ | ✓ |
| Oligoastrocytoma | WHO grade II | ✓ | ✗ |
| Anaplastic Oligoastrocytoma | WHO grade III | ✓ | ✓ |
| Glioblastoma | WHO grade IV | ✓ | ✓ |
Resources to help you feel better resourced and engaged with your clinical care
'Treatment' patient guide
Our 'Treatment' patient guide will guide you through what you can expect at this stage of the pathway. It also suggests some questions you can ask clinicians.
View patient guide

'Diagnosis' patient guide
Our ' Diagnosis' patient guide will guide you through what you can expect at this stage of the pathway. It also suggests some question to ask clinicians.
View patient guide

Who's who in your clinical team
Even before a diagnosis is reached, several people may already have become involved with your situation. This can feel overwhelming and leaving you feeling unsure of who to talk to about different aspects of your care.
The first step to feeling in control when it comes to your clinical care is being able to understand the roles of medical professionals that you may come across following a brain tumour diagnosis.
We have produced our ‘Who’s who in your clinical team‘ guide to help you navigate your pathway and keep a track of essential contacts you need within your clinical team.
Our guide
‘Who’s who in your clinical team’ is our guide to medical professionals who you may meet along your journey. This helpful guide can help you to navigate your pathway and keep track of essential contacts you need within your clinical team.
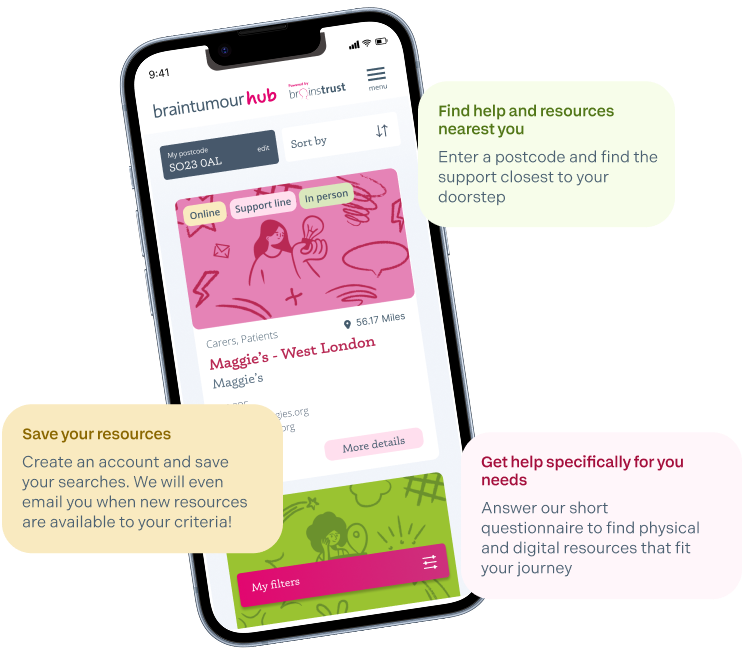
Created for brain tumour patients and caregivers, the brain tumour hub is an easy to navigate database of verified brain tumour support resources that have proven valuable to others.
Find support and resources from across the UK
It has been a delight to collaborate with brainstrust. By referring our patients and their friends and family to this amazing charity we are able to ensure that all aspects of their needs are met.
Your help has been monumental to me in working through my thoughts and understanding of the disease. You have helped me gain a perspective on what has happened and to look at things in a new light. Thank you so very much for all your time and kindness.
Thank you for all your help, you were there when I was at my wits end and both the website and the box gave me the means to take back some control
Once again I can not tell you how reassured you made me feel yesterday. It was so important that I spoke to someone and you were there which I really appreciated
If it wasn't for brainstrust supporting me through my experience, I don't know where I would be right now
Your help has been monumental to me in working through my thoughts and understanding of the disease. You have helped me gain a perspective on what has happened and to look at things in a new light. Thank you so very much for all your time and kindness
Received the brain box yesterday. I cried with happiness. It's just what we needed - the booklets, information, tea and even hand cream. You have restored our faith in human nature

Help fund our mission
improvement in wellbeing for people with a brain tumour thanks to our work.
*See our impact report for a full break down of our impact
by donating only £3 a month, helps us keep this information up to date.



